Linq heart monitor4/6/2023  Failure to follow these conditions for use may result in a hazard to the patient during an MRI scan: Hydrogen proton MRI equipment must have a static magnetic field of 1.5 Tesla (T) or 3.0 T. The Reveal LINQ device can be safely scanned in patients under the following conditions. Medtronic is awaiting FDA decisions on devices including the InterStim Micro sacral nerve stimulator, a treatment for incontinence, and for the MiniMed 770G hybrid closed loop insulin pump system. Reveal LINQ Insertable Cardiac Monitor MRI Conditions for Use. 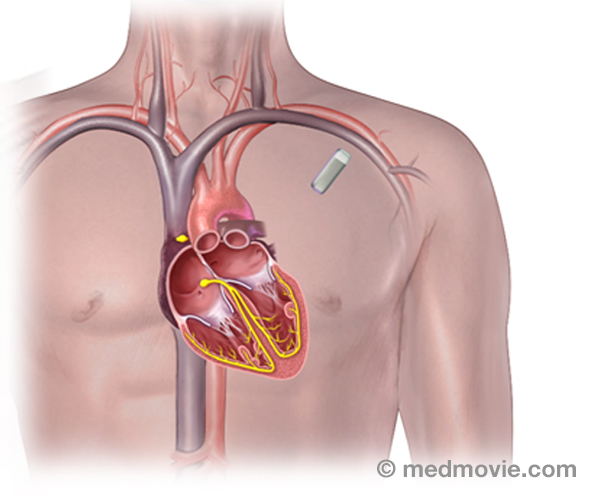 
The company gained FDA approval in June for its Percept deep brain stimulator that can record a patient's brain signals while delivering treatment for conditions such as Parkinson's disease, epilepsy and obsessive-compulsive disorder. Medtronic's Martha said in May that the pandemic did not appear to be affecting the authorization process for products undergoing U.S. Newcomer Boston Scientific has valued the market at about $750 million. In addition to Boston Scientific, Medtronic is also competing against Abbott, with its Confirm Rx system, in the market for insertable cardiac monitors. Medtronic also touted low published rates of false atrial fibrillation detection and a reduction for physicians in time spent reviewing monitor data transmissions as benefits of the new device. The company claims the longevity of its Reveal Linq insertable cardiac monitor is up to three years. 
The latest version fo the device, described as being one-third the size of a AAA battery, has better accuracy in detecting abnormal heart rhythms as well as improved longevity of 4.5 years, Medtronic said. Medtronic said the Linq II system can also detect premature ventricular contraction, which is an extra, abnormal heartbeat that begins in the ventricles and disrupts the heart rhythm. Patient data generated through continuous monitoring can aid physicians in the diagnosis and management of chronic cardiac arrhythmias such as atrial fibrillation. Patients can use their smartphones to automatically transfer device data with a mobile app that enables secure communication via Bluetooth, or they can transmit device data via a home communicator. In addition to remote programming, the Linq II device allows patients to choose between two monitoring avenues. Insertable cardiac monitors (ICMs) are leadless subcutaneous devices that continuously monitor the heart rhythm and record events over a timeframe measured. Medtronic CEO Geoff Martha has called such features critical for devices and asserted that the company is well positioned to take advantage of hospitals' growing demand for the functions, particularly as providers work to reduce their employees' exposure to the virus. © The Author 2015.Remote monitoring and programming capabilities for medical devices have gained a new level of attention in the COVID-19 era by reducing the need for as much in-person interaction with doctors. Published on behalf of the European Society of Cardiology. Hospitals save resources when the higher price of the Reveal LINQ does not outweigh these savings.Ĭatheterization laboratory Costs Implantable cardiac monitor Procedure room Syncope. Inserting the miniaturized version of the ICM is simpler and faster, and the procedure can take place outside the cath lab in a less resource intensive environment. The patient care pathway can be improved due to the possibility to move the procedure out of the cath lab. The net effect on savings depends on the price differential between these two technologies. These cost savings were primarily realized through fewer staff, less equipment, and overhead costs. a Reveal LINQ insertion outside a cath lab resulted in an estimated reduction of €662 for the UK, €682 for the Netherlands, and €781 for France. The cost comparison of a Reveal XT implantation in a cath lab setting vs. Hospitals in the Netherlands, France, and the UK were included in this study. Data were collected from interviews with physicians, cath lab managers, and financial controllers. This study aims to assess the change in procedure costs when performed outside the cath lab.Ī bottom-up costing methodology was used. The procedure is therefore minimally invasive and can be moved from catheterization laboratory (cath lab) to a less resource intensive setting. The newest ICM, Reveal LINQ™ (Medtronic Inc.), is miniaturized and inserted with a specific insertion tool kit. to diagnose unexplained syncope or for detection of suspected atrial and ventricular arrhythmias. 
Implantable cardiac monitors (ICMs) are used for long-term heart rhythm monitoring, e.g. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
